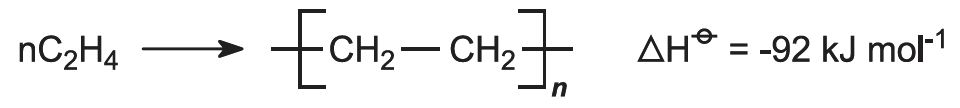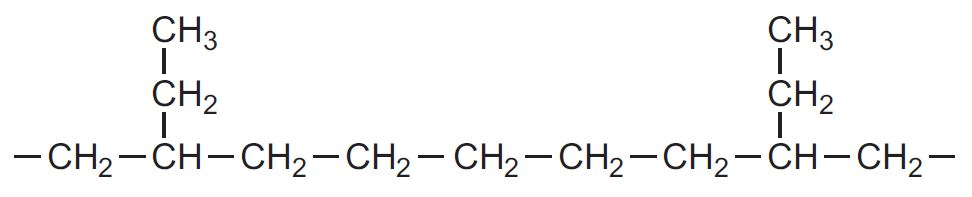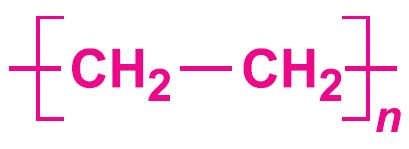
Uses of poly(ethene) (polyethylene)
Poly(ethene) is produced in three main forms: low density (LDPE) (< 0.930 g cm-3) and linear low density ( LLDPE) (ca 0.915-0.940 g cm-3) and high density (HDPE) (ca 0.940-0.965 g cm-3).
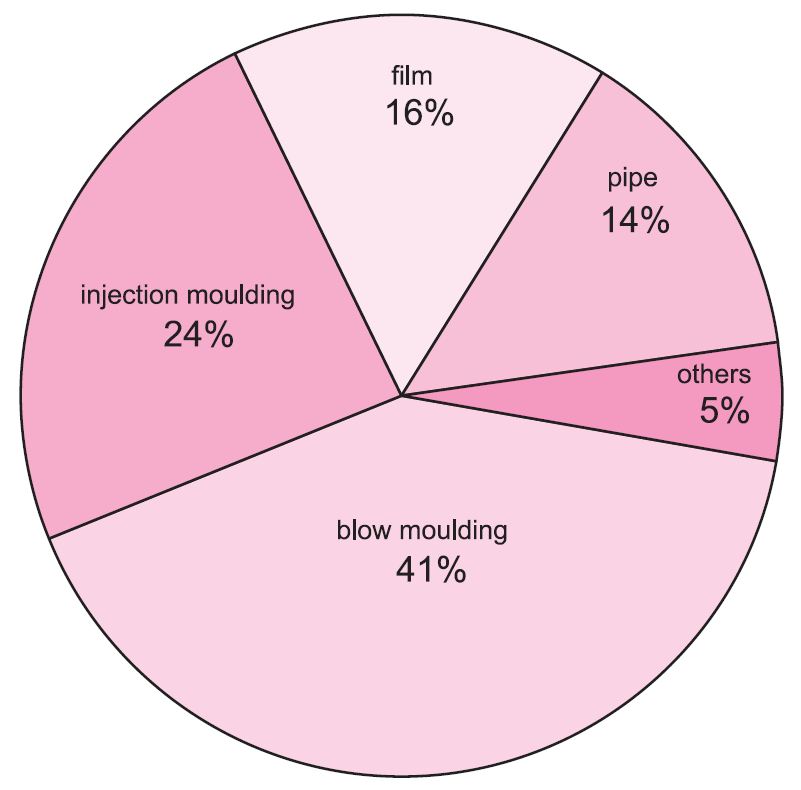
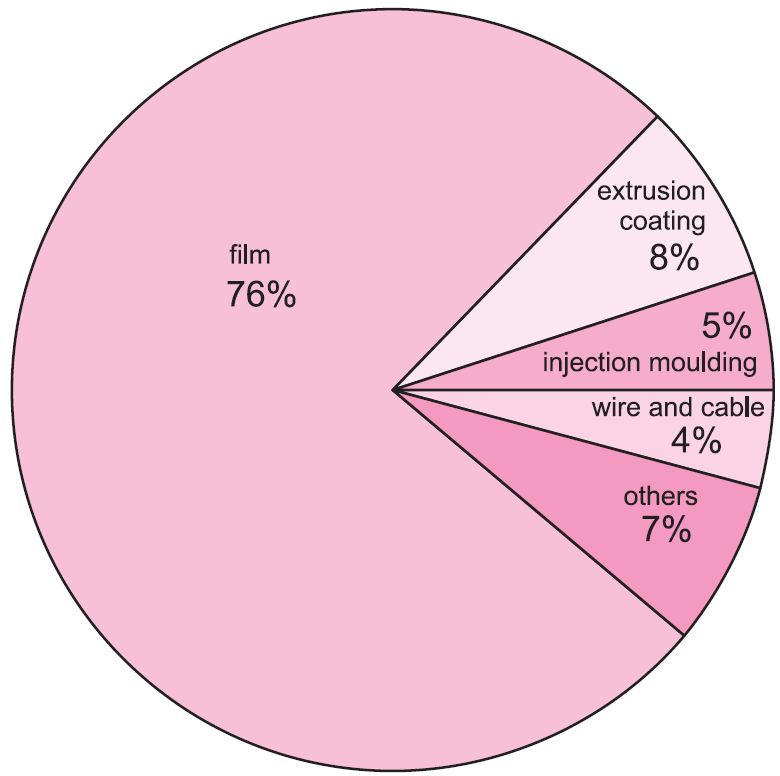
The LDPE or LLDPE form is preferred for film packaging and for electrical insulation. HDPE is blow-moulded to make containers for household chemicals such as washing-up liquids and drums for industrial packaging. It is also extruded as piping.
HDPE
LDPE/LLDPE
Figure 1 Uses of poly(ethene).
All forms can be used for injection-moulded products such as buckets, food boxes and washing-up bowls (Table 1).
| Process | HDPE | LDPE | LLDPE |
|---|---|---|---|
| Making film | Food packaging Shopping bags |
Cling film Milk carton lining |
Stretch film |
| Injection moulding | Dustbins Crates |
Buckets Bowls |
Food boxes |
| Blow moulding | Detergent bottles Drums |
Squeezable bottles | |
| Extrusion | Water pipes | Flexible water pipes Cable jacketing |
Cable coating |
Table 1 Examples of uses of poly(ethene).
|
||||
Annual production of poly(ethene) (polyethylene)/million tonnes1
| In 2013, 2015 | 2018 (est) | |
|---|---|---|
| World | 81.8 | 99.6 |
| North America2 | 16.0 | 18.1 |
| Europe3 | 12.9 | 13.8 |
| Asia Pacific | 36.6 | 47.5 |
| Others | 16.3 | 20.2 |
1. Freedonia, 2014
2. US: 17.4 million tonnes in 2014. 2015 Guide to the Business of Chemistry, American Chemistry Council
3. 14.0 million tonnes in 2015, Plastics - the Facts 2016 PlasticsEurope 2016
| LDPE | LLDPE* | HDPE* | |
|---|---|---|---|
| World4 | 18.7 | 24.1 | 37.5 |
| US5 | 3.2 | 6.3 | 7.9 |
| Europe6 | 8.27 | 5.8 | |
4. Nexant and ChemVision, 2014
5. 2015 Guide to the Business of Chemistry, American Chemistry Council
6. Plastics- the Facts 2016, PlasticsEurope, 2016
7. LDPE plus LLDPE
* Many plants can produce both forms of poly(ethene) and alter the amount that they produce of each type at short notice. Both use a Ziegler (or Phillips) catalyst. If pure ethene is used, HDPE is formed. LLDPE is produced when a small amount of another alkene, for example but-1-ene, is added to the ethene.
Another form, discussed below, mLLDPE, is, at present, produced in much smaller quantities.
Manufacture of poly(ethene) (polyethylene)
Poly(ethene) is made by several methods by addition polymerization of ethene, which is principally produced by the cracking of ethane and propane, naphtha and gas oil.
A new plant is being constructed in Brazil for the production of poly(ethene), from ethene, that is made from sugar cane via bioethanol. This is sometimes termed bio-based poly(ethene) (bio-based polyethylene).
Low density poly(ethene) (LDPE)
The process is operated under very high pressure (1000-3000 atm) at moderate temperatures (420-570 K) as may be predicted from the reaction equation:
This is a radical polymerization process and an initiator, such as a small amount of oxygen, and/or an organic peroxide is used.
Ethene (purity in excess of 99.9%) is compressed and passed into a reactor together with the initiator. The molten poly(ethene) is removed, extruded and cut into granules. Unreacted ethene is recycled. The average polymer molecule contains 4000-40 000 carbon atoms, with many short branches.
For example,
It can be represented by:
There are about 20 branches per 1000 carbon atoms. The relative molecular mass, and the branching, influence the physical properties of LDPE. The branching affects the degree of crystallinity which in turn affects the density of the material. LDPE is generally amorphous and transparent with about 50% crystallinity. The branches prevent the molecules fitting closely together and so it has low density.
High density poly(ethene) (HDPE)
Two types of catalyst are used principally in the manufacture of HDPE:
- a Ziegler-Natta organometallic catalyst (titanium compounds with an aluminium alkyl).
- an inorganic compound, known as a Phillips-type catalyst. A well-known example is chromium(VI) oxide on silica, which is prepared by roasting a chromium(III) compound at ca 1000 K in oxygen and then storing prior to use, under nitrogen.
HDPE is produced by three types of process. All operate at relatively low pressures (10-80 atm) in the presence of a Ziegler-Natta or inorganic catalyst. Typical temperatures range between 350-420 K. In all three processes hydrogen is mixed with the ethene to control the chain length of the polymer.
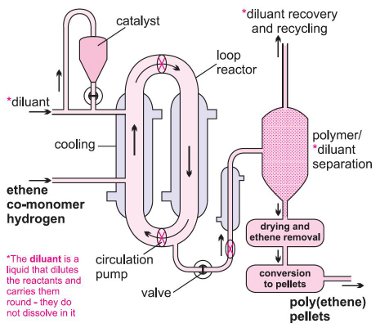
(i) Slurry process (using either CSTR (continuous stirred tank reactor) or a loop)
The Ziegler-Natta catalyst, as granules, is mixed with a liquid hydrocarbon (for example, 2-methylpropane (isobutane) or hexane), which simply acts as a diluent. A mixture of hydrogen and ethene is passed under pressure into the slurry and ethene is polymerized to HDPE. The reaction takes place in a large loop reactor with the mixture constantly stirred (Figure 4). On opening a valve, the product is released and the solvent is evaporated to leave the polymer, still containing the catalyst. Water vapour, on flowing with nitrogen through the polymer, reacts with the catalytic sites, destroying their activity. The residue of the catalyst, titanium(IV) and aluminium oxides, remains mixed, in minute amounts, in the polymer.
|
|
| Figure 5 The slurry process using a loop reactor. By kind permission of Total. |
Figure 4 The manufacture of poly(ethene) using the
slurry process in a loop reactor.
(ii) Solution process
The second method involves passing ethene and hydrogen under pressure into a solution of the Ziegler-Natta catalyst in a hydrocarbon (a C10 or C12 alkane). The polymer is obtained in a similar way to the slurry method.
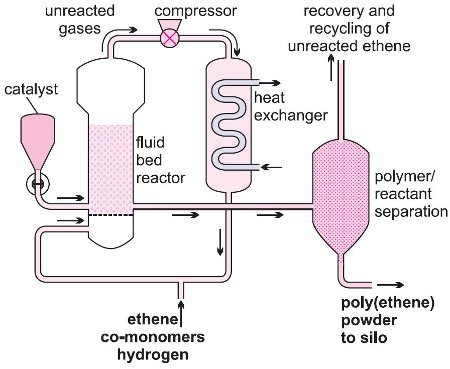
(iii) Gas phase process
Figure 6 Low pressure gas-phase process.
A mixture of ethene and hydrogen is passed over a Phillips catalyst in a fixed bed reactor (Figure 6).
Ethene polymerizes to form grains of HDPE, suspended in the flowing gas, which pass out of the reactor when the valve is released.
Modern plants sometimes use two or more of the individual reactors in series (for example two or more slurry reactors or two gas phase reactors) each of which are under slightly different conditions, so that the properties of different products from the reactors are present in the resulting polymer mixture, leading to a broad or bimodal molecular mass distribution. This provides improved mechanical properties such as stiffness and toughness.
| Figure 7 Granules of poly(ethene) which are then used to make film, extruded into pipes or moulded. By kind permission of Total. |
 |
The HDPE powder coming out of any of the reactors discussed above is separated from the diluent or solvent (if used) and is extruded and cut up into granules.
This method gives linear polymer chains with few branches. The poly(ethene) molecules can fit closer together. The polymer chains can be represented thus:
This leads to strong intermolecular bonds, making the material stronger, denser and more rigid than LDPE. The polymer is not transparent.
Linear low density poly(ethene) (LLDPE)
Low density poly(ethene) has many uses but the high pressure method of manufacture by which it is produced has high capital costs. However, an elegant technique has been developed, based on both Ziegler-Natta and inorganic catalysts to produce linear low density poly(ethene) LLDPE, which has even improved properties over LDPE. Any of the three processes, slurry, solution and gas phase, can be used when a Ziegler-Natta catalyst is chosen. The gas phase process is used when the inorganic catalyst is employed.
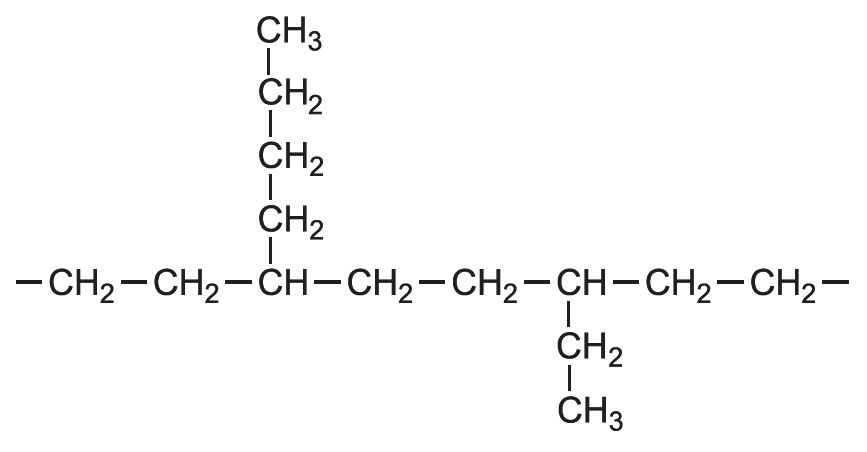
Small amounts of a co-monomer such as but-1-ene or hex-1-ene are added to the feedstock. The monomers are randomly polymerized and there are small branches made up of a few carbon atoms along the linear chains.
For example, with but-1-ene, CH3CH2CH=CH2, the structure of the polymer is:
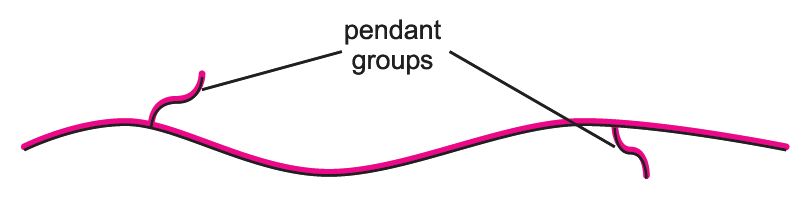
The side chains are known as pendant groups, or short chain branching. The molecule can be represented as:
The structure is essentially linear but because of the short chain branching it has a low density. The structure gives the material much better resilience, tear strength and flexibility without the use of plasticisers. This makes linear low density poly(ethene) an ideal material for the manufacture of film products, such as those used in wrappings.
The properties of the polymer, and hence its uses, can be varied by varying the proportion of ethene and co-monomer and by using different co-monomers. All this can be done without shutting down the plant, an enormous advantage.
Metallocene linear low density poly(ethene) (mLLDPE)
|
|
| Figure 8 Poly(ethene) film is used extensively for wrapping foods. By kind permission of BP. |
This poly(ethene), known as mLLDPE, is produced by a new family of catalysts, the metallocenes. Another name for this family is single site catalyst. The benefit is that the mLLDPE is much more homogenous in terms of molecular structure than classical LLDPE produced by Ziegler-Natta catalysts. Each catalyst is a single site catalyst which produces the same PE chain. Chemists have compared the structure of metallocenes to that of a sandwich. There is a transition metal (often zirconium or titanium) 'filling' a hole between layers of organic compounds.
The catalysts are even more specific than the original Ziegler-Natta and it is possible to control the polymer's molecular mass as well as its configuration. Either the slurry or solution processes are usually used.
Poly(ethene) produced using a metallocene can be used as very thin film which has excellent optical properties and sealing performance, thus making them very effective for wrapping foods. The real plus for the metallocene catalysts are the enhanced mechanical properties of the films made out of mLLDPE.
Co-polymers
Ethene forms co-polymers with propene which have very useful properties.
Date last amended: 27th April 2017