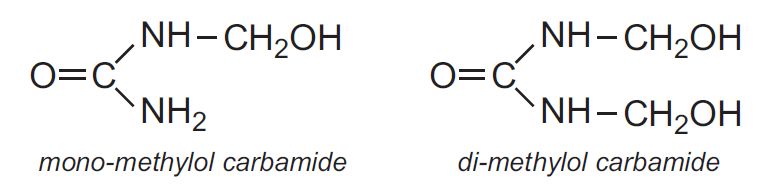Methanal (formaldehyde) reacts with phenol, carbamide (urea) and melamine to form three of the most important thermosetting plastics (resins). Sometimes, the resins are called PF (phenol-formaldehyde), UF (urea-formaldehyde) and MF (melamine-formaldehyde). The processes are all examples of condensation polymerization.
Annual, world production of methanal plastics 1
| Phenol-methanal plastics | 16 million tonnes |
| Carbamide-methanal plastics | 40 million tonnes |
| Melamine-methanal plastics | 21 million tonnes |
1. These data for 2015 have been estimated from Research and Markets 2012 and MMSA, 2016
Phenol-methanal (phenol-formaldehyde) plastics
These are amongst the oldest plastics, Backland patenting the process in 1910. They are prepared from phenol and methanal on the addition of acid or alkali.
Manufacture of phenol-methanal plastics
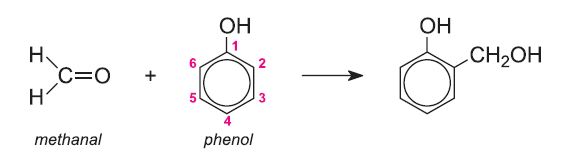
Phenol and methanal are heated in an acidic or alkaline solution.
The first step in forming a polymer chain involves substitution of methanal in the phenol ring in the 2- or 4- position:
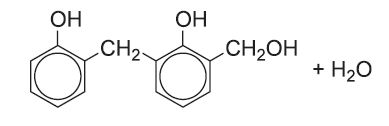
The product then undergoes a condensation reaction:
When methanal and phenol, in the proportion of 0.5:1, are reacted under acidic conditions, a resin (often known as a novalac resin) is produced, where n = 0 to 6.
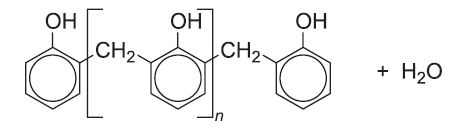
With higher proportions of methanal and with the 4-position vacant, branching is possible, the methanal providing additional -CH2- links. On heating a resin is produced. Part of the 3-dimensional cross-linked structure is illustrated:
The properties of a resin can be altered by using other phenols. For example, if the phenol has an alkyl group with four or more carbon atoms (for example, a butyl group) in the 4-position, the resulting polymer is soluble in oils. Such polymers also have lighter colours.
Uses of phenol-methanal plastics
The polymers with giant three-dimensional, cross-linked structures form resins that cannot be melted and reshaped. On heating to high temperatures, they begin to decompose and char. They are also insoluble in a wide range of solvents and are unreactive. Their high electrical and heat resistance means that they are used as an electrical insulator and for the manufacture of brake and clutch linings for vehicles. When they are used with fillers such as wood flour (wood that has been pulverised roughly to the size of a rice grain) or mica, they have major applications in the construction of worktops and printed circuit board insulation. They are also widely used as adhesives, for example in producing plywood and hardboard.
Carbamide (urea)-methanal (formaldehyde) plastics
Of the three methanal plastics, the carbamide (urea) - methanal (formaldehyde) resins (UF) are the most used.
Manufacture of carbamide-methanal plastics
Carbamide (urea) is manufactured by heating ammonia and carbon dioxide under pressure. Reaction between methanal and carbamide (urea) in alkaline solution gives mono- and di-methylol carbamides:
These products condense together in acidic conditions to give a linear polymer. Water is eliminated:
Further heating (curing) causes further condensation, with the elimination of water between the OH and NH groups, resulting in a cross-linked 3-dimensional structure.
The plastic is produced either as a powder or as granules.
Uses of carbamide-methanal plastics
These plastics are often used after incorporating fillers such as paper pulp and wood flour. They have the advantage over phenolic resins of being colourless, although they are less water resistant and less heat resistant.
Their greatest use is as wood adhesives, usually mixed at point of use with filler, extenders and acidic hardeners. They are used in particular as a binder in the production of chipboard. The resin powder or granules are moulded into electrical plugs and sockets, toilet seats and some table wear. Used in solution, the resins wet-strengthen paper and improve the shrink resistance of cotton, wool and rayon where a stiff finish is required.
The key restraint is the emission of toxic emissions of methanal vapour which has been of concern in many countries. For example, the California Air Resources Board has strict limits on the amount of methanal emitted and this has restricted the use of the resin.
Melamine-methanal (melamine-formaldehyde) plastics
Melamine resins combine the best of the properties of phenolic (PF) and carbamide (urea) (UF) resins. They are stable to heat, light and moisture and are colourless, and can absorb dyes easily.
Manufacture of melamine-methanal plastics
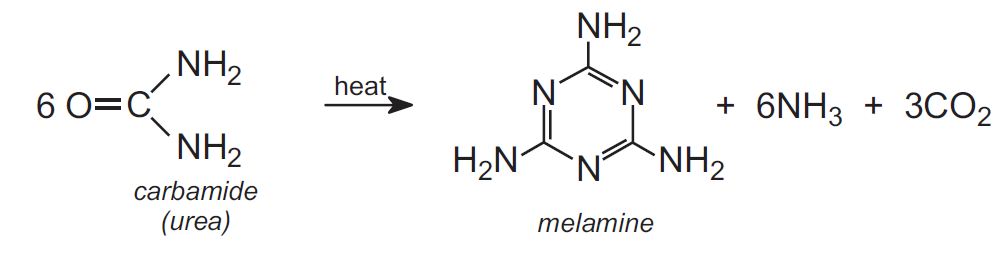
Melamine is produced by heating carbamide (urea):
The production of melamine-methanal (MF) plastics is similar to that for carbamide (urea)-methanal polymers, and their structures are analogous.
The resin is usually produced as a powder.
Uses of melamine-methanal plastics
The powder can be moulded under pressure and most of the resin is used to make laminates for surfaces. Kitchen worktops can be made using a cheaper phenolic resin to form the base, which is then coated with a more expensive melamine surface. Light-weight picnicware and tableware (e.g. Melaware) can also be made from melamine.
Like the carbamide (urea) resins, melamine resins are also used, in solution, to wet-strengthen paper and to improve the shrink resistance of cotton, wool and rayon. Resins are also used as adhesives and as ion-exchange resins to demineralise water. If melamine, methanal and an alcohol (for example, butanol) are heated together, a resin is formed with both -CH2OH and -CH2OR groups. The resulting resins are then cross linked with other polymers to form paints for cars.

Figure 1 To absorb sound, and make commuting more comfortable, the roofs of
25 new subway stations built recently in Seoul, Republic of Korea are constructed of
perforated steel behind which is a layer of melamine resin. The resin is produced as
three-dimensional network structure consisting of slender and thus easily shaped webs.
By kind permission of BASF.
Date last amended: 9th May 2017