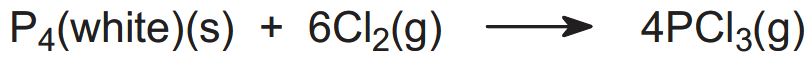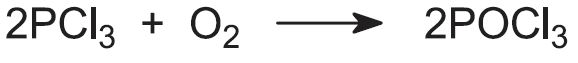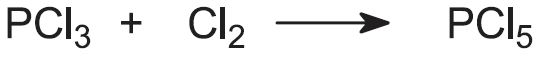Elemental phosphorus (frequently called white (or yellow) phosphorus, P4) is only required for the production of a limited number of chemicals and most phosphorus consumed worldwide is consumed in the form of phosphates.
Uses of phosphorus
There are various allotropic forms of phosphorus but only two forms have commercial significance - white and red phosphorus - and white phosphorus is the most commercially important, accounting for 99% of demand worldwide.
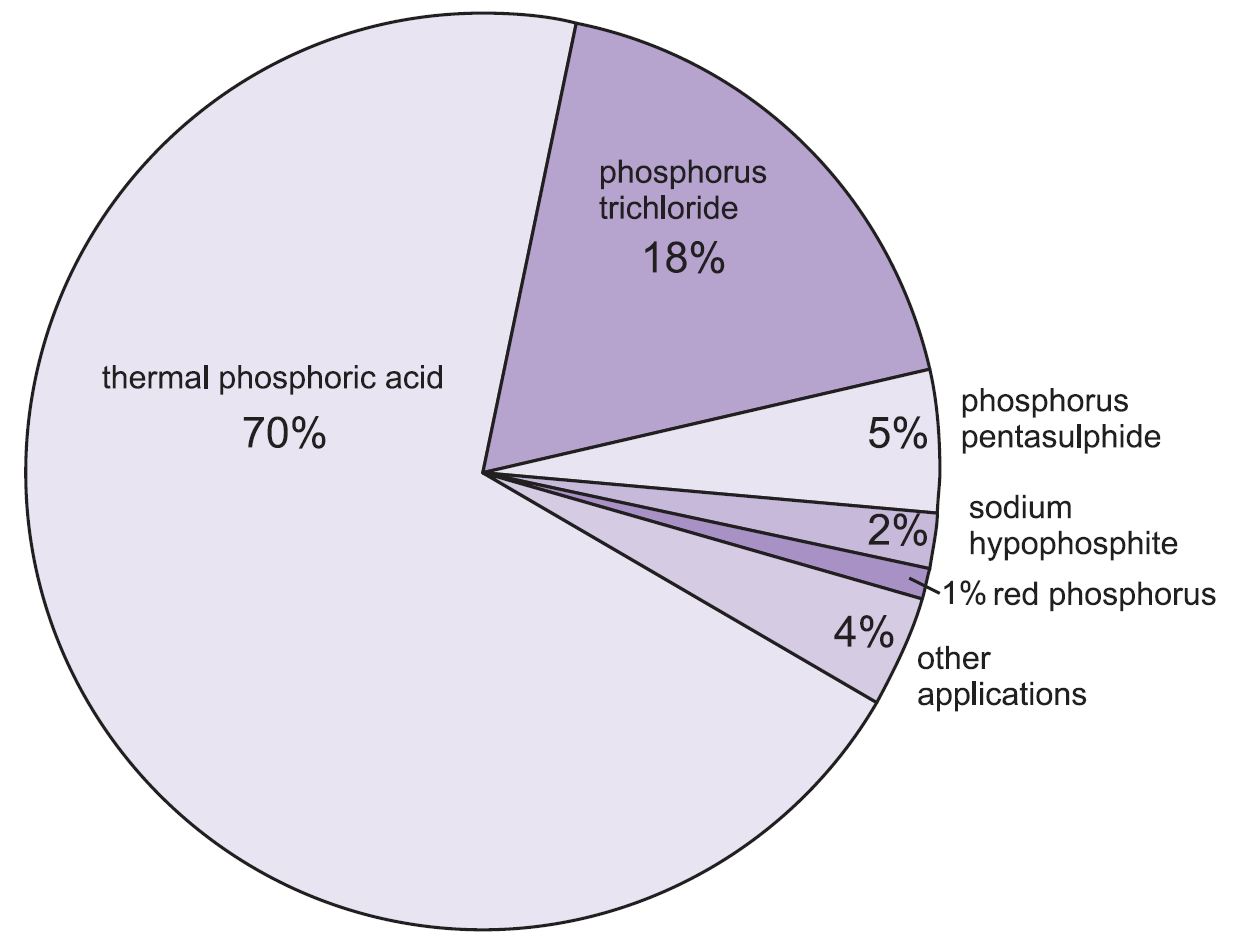
Most phosphorus is used to make phosphoric acid of high purity necessary for some food processing and for etching semi-conductors. It is known as thermal phosphoric acid and is made by burning phosphorus in moist air.
However most phosphoric acid needed to make fertilizers need not be very pure and is more cheaply made directly from rock phosphate ores and not via the element.
Elemental phosphorus is also used to manufacture other chemical intermediates (products used in the manufacturing processes for other chemical products).
Figure 1 Uses of phosphorus.
The uses of some of the key phosphorus compounds, such as phosphorus trichloride, phosphorous acid, the phosphorus sulfides, sodium hypophosphite, phosphine and the phosphides are discussed in this unit.
Phosphoric acid and the phosphates are very widely used.
Red phosphorus is used in pyrotechnics and matches. It is also used as a flame retardant in plastics (particularly polyamides) where its rapid oxidation consumes all the oxygen present, thereby stopping the fire.
Annual production of elemental phosphorus
Data for the production of phosphorus in terms of tonnage are not available. It is thought that China has about 84% of the global capacity, followed by Kazakhstan, the US and Western Europe at 6%, 5% and 5% respectively1.
1. IHS Markit, 2013
Manufacture of white phosphorus
Elemental phosphorus production is by an electrothermal process and consequently the energy demand is very high. Each tonne of phosphorus produced requires about 14 MWh. Its manufacture is carried out only where comparatively cheap energy, such as hydroelectric power, is available. The main producers are in Kazakhstan, China and the United States.
The most important source is fluoroapatite, 3Ca3(PO4)2.CaF2 commonly known as 'phosphate rock'. Impurities present include calcium and magnesium carbonates, iron(lll) oxide, aluminium oxide, and silica. It occurs widely but important sources are in the US (Florida, North Carolina), Morocco, Tunisia, Russia and China.
To optimize production efficiency, the feed of phosphate rock into a furnace needs to be as uniform as possible. To achieve this, phosphate rock is heated strongly in an oven (usually a rotating calciner) which results in the fusion of small particles to form the hard nodules required for satisfactory furnace operation. The furnace, containing three vertical carbon electrodes, is fed with a mixture of coke, sand and phosphate rock in a mass ratio of, typically, 16:30:100.
A modern furnace has a diameter of approximately 12 m and is 8 m high and can produce 30 000 tonnes of phosphorus a year.
The reaction may be represented as:

Gaseous phosphorus and carbon monoxide, from the top of the furnace, are passed into a spray of water at 343 K. Most of the phosphorus (mp 317 K) condenses. Condensation is completed using cold water.
The carbon monoxide is either burnt off or recycled as a source of fuel. It can be used in the production process for the preparation of the nodules from the phosphate rock, or in some instances is sold to local power producers.
Molten calcium silicate slag and an alloy of iron and phosphorus, known as ferrophosphorus are removed separately from the base of the furnace. The ferro-phosphorus can be added to sheet steel to prevent sticking of sheets or added to iron to improve stiffening. This improves machineability in certain steels. Some calcium silicate is used as low-grade hardcore.
White phosphorus ignites spontaneously in air, and therefore is stored under warm water as a lower liquid layer. During storage, a phosphorus/water/solids mixture accumulates at the water surface and this 'phosphorus mud' is either pumped back to the furnace for recovery of the phosphorus or distilled in separate 'mud' distillation units. The 'phossy water' arising from the condensers can contain dissolved and suspended elemental phosphorus, as well as phosphorus oxy-acids, hexafluorosilicic acid, ammonia, and silica. Phossy water is neutralized with lime (occasionally ammonia), settled, and then treated with chlorine to remove the last traces of phosphorus.
Phosphorus is transported in tanks under water, or under nitrogen, the tanks being heated by hot water pipes. UK supplies are imported in specially designed rail cars carrying approximately 50 tonnes of the solid element, which has to be melted before discharge. It is melted with hot water (indirect heating) then kept at 330 K in the rail car and discharged directly from the rail car to a storage tank where it is stored underwater.
Manufacture of red phosphorus
Red phosphorus, unlike white phosphorus, is not spontaneously flammable, although it is easily ignited.
It is manufactured from white phosphorus. White phosphorus is run from its storage tank into a steel pot where it is kept under a layer of water. A lid, fitted with a safety pipe, is securely fastened and the pot is heated to 550 K for 3 to 4 days. The water escapes as steam through the safety pipe and phosphorus vapour loss is prevented by a reflux condensing system.
After 48 hours the temperature is raised to 673 K when most of the unchanged white phosphorus distils off (bp 553 K).
The pot contents are kept wet and ground to give a slurry of red phosphorus. After decanting most of the water, sodium carbonate is added. On boiling, the remaining white phosphorus is destroyed.
Finally, the red phosphorus is removed and vacuum dried.
Phosphorus compounds
(a) Phosphorus trichloride
The total volume of phosphorus trichloride manufactured each year is about 700 000 tonnes, by the direct reaction between phosphorus and chlorine:
It is highly reactive to atmospheric moisture and is transported in lead-lined, glass-lined or nickel vessels.
Phosphorus trichloride has a number of important applications which include the production of:
- phosphorus oxychloride (POCl3)
This is done by direct reaction with oxygen:
POCl3 is used as a chlorinating agent.
- phosphorous acid (H3PO3)
In recent years the most important application of PCl3 has been the production of the herbicide glyphosate, which requires that it is first converted to phosphorous acid (see below).
- additives for the plastics, lubrication oils, paints, and other industries
- phosphorus pentachloride (PCl5)
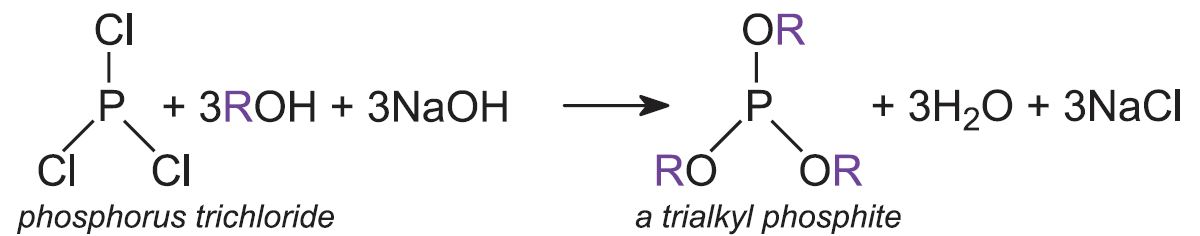
- trialkyl phosphites Trimethyl phosphite is an important example which is used to make organophosphorus herbicides, pesticides and flame retardants.
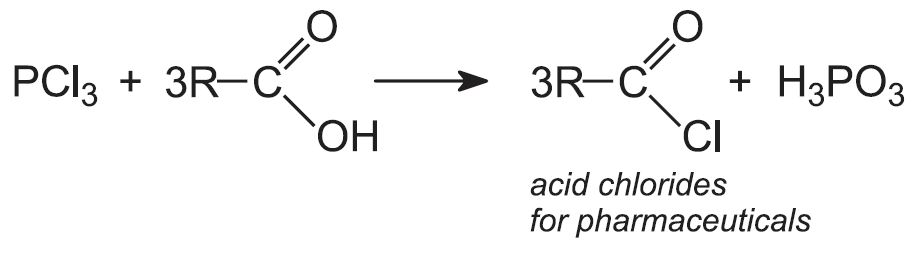
- acid chlorides
These are used in pharmaceutical synthesis
(b) Phosphorous Acid
This is made by hydrolyzing phosphorus trichloride in dilute phosphorous acid. The acid is concentrated in evaporators and the molten product cooled. It solidifies into flakes.
Phosphorous acid is used in the manufacture of herbicidal glyphosates.
Glyphosate is the world's most widely used and commercially valuable chemical for crops. It is produced by heating a mixture of phosphorous acid with glycine (2-aminoethanoic acid) and then adding methanal.
(c) Phosphorus sulfides
White phosphorus reacts with sulfur to form a variety of sulfides, P4S10, P4S7, P4S5, P4S3. By far the most important of these is P4S10, which is normally referred to as phosphorus pentasulfide, P2S5. It is used to make lubricating oil additives which act as antioxidants, corrosion inhibitors and antiwear additives, and as an intermediate in the manufacture of insecticides such as sulfur-based organophosphorus pesticides.
There are over 52 registered pesticides for which P2S5 is an intermediate. It is reacted with either methanol, ethanol, or propan-2-ol (isopropanol) to produce respectively dimethylphosphorodithioic acid (DMPA), diethylphosphorodithioic acid (DEPA), or diisopropylphosphorodithioic acid (DIPA). These are key intermediates for the organophosphate pesticide industry.
Small amounts of phosphorus sesquisulfide, P4S3, are used in the match industry. However the product is only used for "strike anywhere" (i.e. non-safety) matches, and the worldwide market for P4S3 is in the region of 100 tonnes per annum. There is only one producer of the product.
(d) Sodium hypophosphite
Sodium hypophosphite monohydrate (SHPM) is the sodium salt of hypophosphorous acid HOP(O)H2. First the calcium salt is made by reacting white phosphorus with a slurry of lime at the boil. Subsequently, the calcium salt reacts with sodium sulfate to form SHPM. This has a number of commercial applications, but the most important is for electroless nickel plating (ENP), in which it acts as a reducing agent. ENP accounts for around 90% of demand. A major growth area for ENP has been the manufacturing of hard disc drives for computers.
(e) Phosphine
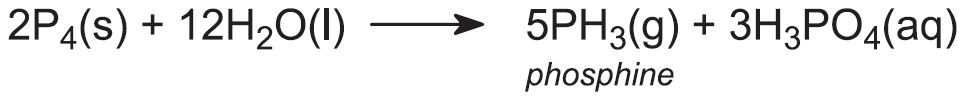
White phosphorus is heated under pressure in water, acidified by phosphoric acid, at about 550 K to yield phosphine:
White phosphorus forms the red allotrope prior to further reaction. The reaction is carried out in graphite batch reactors.
About 1500 tonnes of phosphine, which is a poison, are made each year and are used to make flame proofing derivatives and for pest control. For example the gas is used in large grain silos, to protect the grain.
(f) Phosphides
Red phosphorus is heated with powdered metals to give phosphides such as aluminium phosphide, AlP, zinc phosphide, Zn3P2, and calcium phosphide, Ca3P2.
Their principal use is to kill rodents such as rats and as a fumigant for stored cereal grain.
The phosphides react with moisture, hydrolyzing to form phosphine and thus they have to be handled very carefully.
Date last amended: 6th January 2017